Hello friends, today we will know what is the quantum number. How many types are there, and their definitions will also be known step by step in easy language, so let’s start?
What is the definition of the quantum number?
Quantum Number –
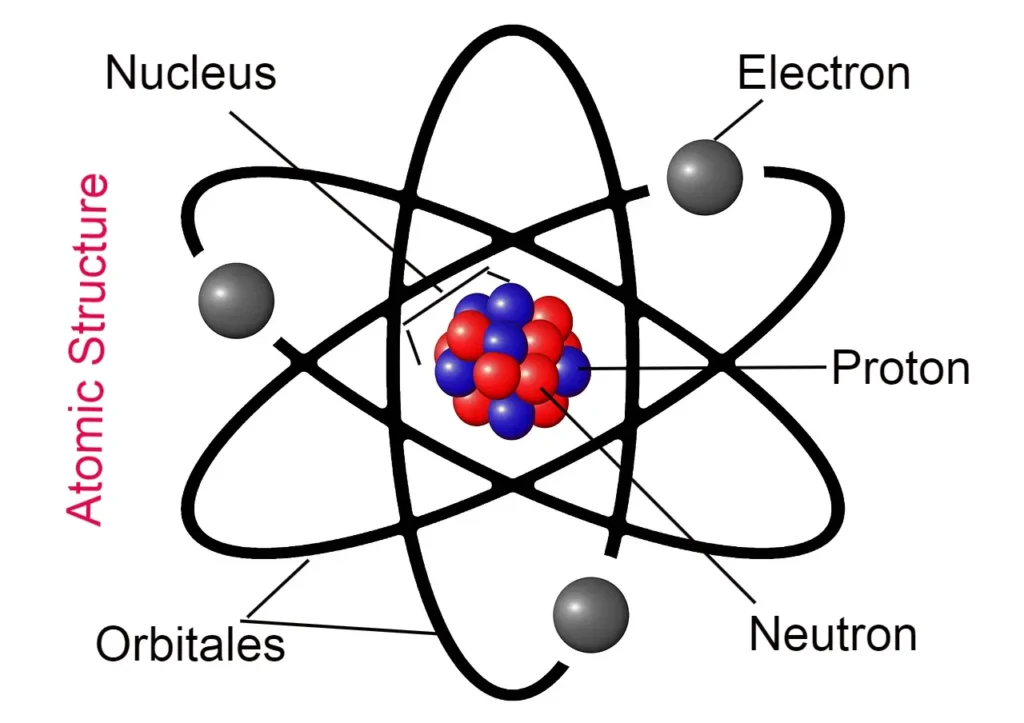
The numbers from which information about the energy and position of the electron in any atom are obtained are called quantum numbers, four types of quantum numbers completely introduce the electron.
How many Types of Quantum Numbers?
There are four types of quantum numbers.
- Principal quantum number
- Azimuthal quantum number
- Magnetic quantum number
- Spin quantum number
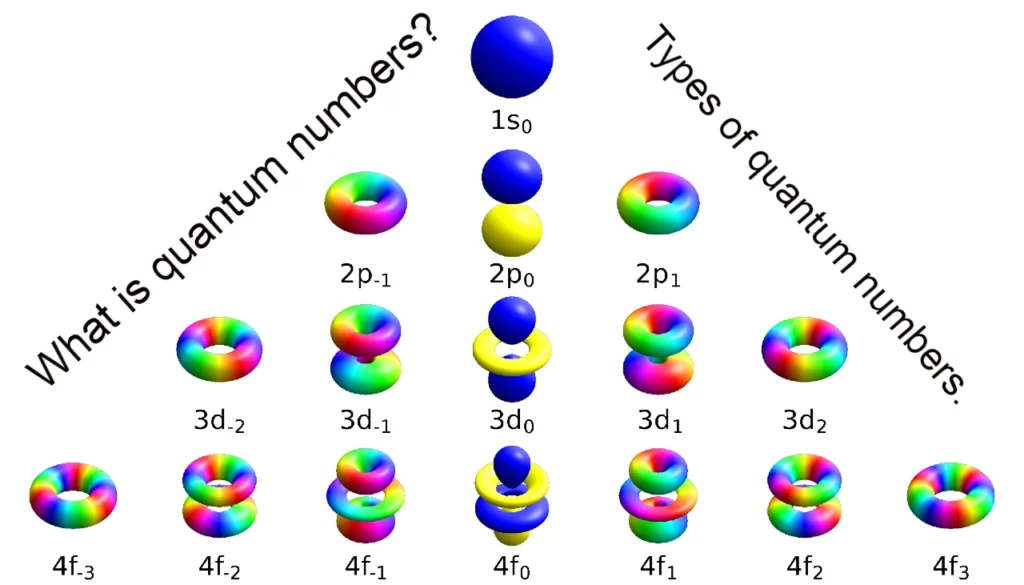
What is the Principal Quantum Number?
- The principal quantum number is denoted by “n”.
- The principal quantum number determines the size of an atom.
- Expresses the atomic nucleus distance of the electron, that is, the atomic radius.
- Determines the velocity of electrons.
- The principal quantum number tells the energy of the electron.
- The core quantum had its origin in the Bohr model.
- It determines the angular momentum of electrons in orbits.
- The principal quantum number tells the amplitude of the wave.
- The value of the principal quantum number ranges from 1 to infinity.
- The spectrum originated in the Bohr model.
What is Azimuthal Quantum Number?
- The azimuthal quantum number is denoted by “l”.
- Provides information about binomial quantum number subshell.
- l = 0 = s subshell
- l = 1 = p subshell
- l = 2 = d subshell
- l = 3 = f subshell
- Expresses angular momentum in an azimuthal quantum number subshell.
- The binomial quantum number represents the frequency of the wave of an electron.
- The value of a binomial quantum number ranges from 0 to (n-1)
- Example – n = 4, l = 0, 1, 2, 3
What is Magnetic Quantum Number?
- The magnetic quantum number is denoted by ‘m’.
- The magnetic quantum number is called the number of orbitals, it represents the number of orbitals.
- The magnetic quantum number represents the configuration of electrons in the three-dimensional region or in space.
- The value of the magnetic quantum number ranges from -l to +l.
- Example-
- l = 1 = p – subshell
- m = -1, 0 +1
What is Spin Quantum Number?
- It is denoted by ‘s’.
- It shows the direction of spin of the electron.
- If the electron spins clockwise, then s will be +1/2; if the electron spins counterclockwise, it will be -1/2.
S = n/2
n = Number of unpaired electrons
n = 3
s = n/2
s = 3/2
If a species has an unpaired electron, then its magnetic moment is found to be
µ = mu = magnetic moment = magnetic moment
µ = under road n(n+2)
µ = (4s(s+1)
The order of quantum numbers is n, l, m, and s, which means that the principal quantum number determines the value of the remaining three quantum numbers while the remaining three quantum numbers cannot determine the value of n.
n = n
l = n
m = n square
s = 2n square
n = 2
l = 0, 1
m = n square = 4
s = 8
For the value of l –
Ex – l = l l = 1
m = (2l+1) m = 3
s = 2 (2l+1) s = 6
Pauli’s Exclusion Principle –
According to Pauli’s exclusion law, no two electrons in an atom can have the same value as all four quantum numbers. The value will be different, i.e. +1/2 for the first electron and -1/2 for the second electron.
Example – According to the law of exclusion of Pauli’s, there are a total of 32 electrons in the fourth main energy level, the number of electrons in the main energy level is 2n2
What is Hund’s Principle?
The distribution of electrons in an orbital is done according to Hund’s law, according to this law, electrons in each orbital of any subshell cannot be paired unless each orbital is unpaired, this is Hund’s law.
Friends, I hope that you have liked the given information about quantum numbers, friends, if you have liked this information, then please share it with your friends as much as possible so that they can also benefit from it.
Thank you very much